
Plasma assays combining p-tau217 and Aβ42/40 accurately confirm AD, supporting patient eligibility for anti-amyloid disease-modifying therapies.

Digital Screening Tool Duo Shows Promise for ADRD Detection in Primary Care

Axsome Submits sNDA for AXS-05 to Treat Agitation in Alzheimer Disease

Plasma assays combining p-tau217 and Aβ42/40 accurately confirm AD, supporting patient eligibility for anti-amyloid disease-modifying therapies.

A study reveals that starting benzodiazepines or antipsychotics in hospice patients with dementia significantly increases 180-day mortality, urging careful prescribing.

An 18-year multicohort study found that smokers who quit even in later life showed 20% slower memory decline vs those who continued to smoke.

Roche's Elecsys pTau181 test has a 97.9% negative predictive value to rule out amyloid pathology, which will improve referral decisions and expedite treatment.

As dementia care grows more complex and more promising, the role for front-line clinicians is becoming more nuanced and more essential.
The ReCODE program targets metabolic, infectious, immune, vascular, and toxic exposures, elements that underlie both mood and cognition, study authors said.

The FDA has granted fast track designation to BMS-986446, an anti–microtubule binding region tau antibody in phase 2 trials for early Alzheimer disease.

The European Commission granted marketing authorization for the Lilly mAb based on data from the phase 3 TRAILBLAZER-ALZ 2 and 6 clinical trials.

A study of more than 559 000 adults and 2.4 million genetic samples found that alcohol consumption of any amount increases dementia risk, challenging prior evidence of protective effects from light drinking.

FDA grants Fast Track designation to etalanetug, a promising tau-targeting antibody for Alzheimer disease that could expand treatment options for the neurodegenerative disease.

Among 180+ active AD trials, 15 biological targets span traditional Aβ and tau proteins as well as new pathways in brain metabolism, neuroinflammation, and synaptic health.

Your daily dose of the clinical news you may have missed.

Once-daily blarcamesine was associated with minimal cognitive decline at 48 weeks, with changes comparable to those observed in prodromal aging.

The FDA has cleared Eisai and Biogen’s Leqembi Iqlik, the first at-home subcutaneous autoinjector for maintenance therapy in patients with early Alzheimer disease.

Your daily dose of the clinical news you may have missed.

The protective effect of the diet was strongest in the highest-risk group, those homozygous for the APOE4 gene variant, suggesting that diet may help offset genetic risk.

Risk of dementia was more than 60% lower among adults aged 60 to 69 years with hearing loss who used hearing aids in the first study to highlight early intervention.

Your daily dose of the clinical news you may have missed.
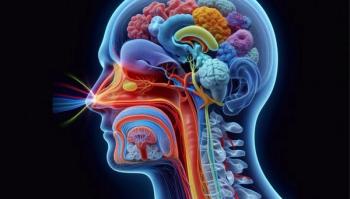
Insulin delivered via nasal spray in this first-in-human PET imaging study was observed in 11 key brain areas relevant to cognitive decline, validating a new research avenue.

Your daily dose of the clinical news you may have missed.

The 2 new plasma assays to enhance Alzheimer’s disease research will support disease staging, predicting disease progression, and monitoring treatment outcome.

Insights on atopic dermatitis treatment preferences, the safety of childhood vaccines, maternal depression’s effect on parenting, and more.


If primary care clinicians are to meet the cognitive screening demands that are growing with an aging population, they need the sharpest tools in the box, this expert says.

A cognitive health assessment will soon be as routine as taking blood pressure or pulse oximetry, says neurocognitive researcher David J. Libon, PhD. He talks more about that future.